contact
Nancy Kamphaus
Administrative Assistant
Do not use this number or leave any messages at this exchange for Oral Medicine Consultants or Oral Pathology Consultants
Professor Meenakshi Chellaiah, PhD
Education and Training
 Unique contributions to Dr. Chellaiah's research projects mark her academic journey. She earned her Ph.D. from the School of Biological Sciences, Madurai University, focused on 'Cardiac Hypertrophy' in rats. Her post-doctoral work at St. Louis University, St. Louis, under the guidance of Dr. Farhang Payvar, and at Washington University, St. Louis, with Dr. Keith Hruska, delved into the characterization of glucocorticoid response elements and bone remodeling processes by bone cells (osteoclasts and osteoblasts), respectively. Her time at Washington University was particularly fruitful, as she secured an R01 grant (1999) on 'Gelsolin-based signaling in osteoclasts and Actin dynamics.' Dr. Chellaiah then held academic positions at the School of Dentistry, University of Maryland, in various departments, including Biomedical Sciences, Oncology and Diagnostic Sciences, Biochemistry & Molecular Biology, and the Program in Oncology at the Marlene and Stewart Greenebaum Comprehensive Cancer Center. With the support of NIH funding for several years (details are below), Dr Chellaiah's laboratory explored the molecular mechanisms of bone resorption and formation by osteoclasts and osteoblasts, respectively. She has been working with bone remodeling since 1991 and cancer-induced bone loss since 2003, demonstrating her extensive experience and expertise in the field.
Unique contributions to Dr. Chellaiah's research projects mark her academic journey. She earned her Ph.D. from the School of Biological Sciences, Madurai University, focused on 'Cardiac Hypertrophy' in rats. Her post-doctoral work at St. Louis University, St. Louis, under the guidance of Dr. Farhang Payvar, and at Washington University, St. Louis, with Dr. Keith Hruska, delved into the characterization of glucocorticoid response elements and bone remodeling processes by bone cells (osteoclasts and osteoblasts), respectively. Her time at Washington University was particularly fruitful, as she secured an R01 grant (1999) on 'Gelsolin-based signaling in osteoclasts and Actin dynamics.' Dr. Chellaiah then held academic positions at the School of Dentistry, University of Maryland, in various departments, including Biomedical Sciences, Oncology and Diagnostic Sciences, Biochemistry & Molecular Biology, and the Program in Oncology at the Marlene and Stewart Greenebaum Comprehensive Cancer Center. With the support of NIH funding for several years (details are below), Dr Chellaiah's laboratory explored the molecular mechanisms of bone resorption and formation by osteoclasts and osteoblasts, respectively. She has been working with bone remodeling since 1991 and cancer-induced bone loss since 2003, demonstrating her extensive experience and expertise in the field.
In addition to her research, she has been a course director for the Cell and Molecular Biology Course and a pre-doctoral director for several years. She has served on master's and PhD thesis committees for several students in their qualifying exams, thesis proposals, and thesis defense processes. Her lab supported the summer research program for high school students, laboratory rotations of several master's and Ph.D. students [GPILs & School of Dentistry research programs], and residents' projects at the Dental School. Besides, she has mentored several graduate and post-doctoral fellows and visiting scholars (list below), further showcasing her leadership and mentorship skills.
Research Projects
-
Projects on Osteoclasts and bone remodeling:
Dr. Chellaiah's first project on Osteoclasts - Osteoclasts (OCs) are bone-resorbing or degrading cells, and bone resorption is their unique function. OCs use podosomes for adhesion and migration and sealing rings for bone resorption. Sealing ring formation has been deemed an indicator of OC activation and bone resorption. Dr. Chellaiah's laboratory dissected the molecular mechanisms behind the cyclical processes of sealing ring formation (Fig. 1). It demonstrated the formation of nascent sealing zones (NSZs) before the mature sealing ring formation. Dr. Chellaiah's laboratory's groundbreaking research identified L-plastin (LPL) as a potential regulator of the actin-bundling process in forming NSZs, precursor zones for mature sealing rings. The genetic deletion of LPL in mice, a significant milestone in her research, demonstrated decreased eroded perimeters and increased trabecular bone density. Moreover, her innovative approach using a TAT-fused cell-penetrating small molecular weight LPL peptide (TAT- MARGSVSDEE, denoted as an inhibitory LPL peptide) attenuated the formation of NSZs and sealing rings (Fig. 2) and impaired bone resorption in vitro in osteoclasts.
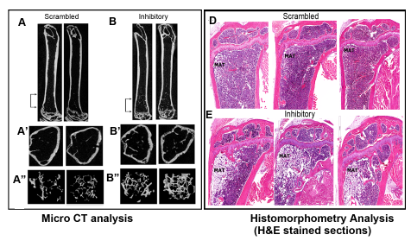
Subsequently, Chellaiah lab injected aging C57BL/6 female mice (36 weeks old) subcutaneously with the inhibitory and scrambled peptides of LPL for 14 weeks. Micro CT and Histomorphometry analyses demonstrated increased trabecular bone density of femoral and tibial bones in mice injected with the inhibitory LPL peptide (Fig. 3). The LPL peptide approach to bone loss is novel. Currently, her lab is testing the impacts of the LPL inhibitory peptide on other diseases that display bone loss due to osteoclast activation. Nanotechnology will be used to deliver the peptides to improve stability and sustain release.
- Patent Award for Inventor Recognition
The inhibitory L-plastin peptide could be a promising drug for improving patient bone density. Dr. Chellaiah was granted a patent for this inhibitory peptide on Nov 1, 2022. The patent title is "Compositions and Methods for Inhibition of L-Plastin Activity in Osteoclasts to Reduce Bone Loss." The US Patent number is 11485762-B2 Chellaiah
Second Project in Osteoclasts. Dr. Chellaiah has developed another project with a graduate student (Dr. Mohammed AlQranei, who graduated in 2020) to determine the C-phycocyanin (C-PC) effect on LPS-regulated osteoclastogenesis. C-PC strongly inhibited the early stage of OC differentiation, thus significantly suppressing RANKL- TNF-alpha- and LPS-mediated osteoclastogenesis. Results from these studies highlighted a possible osteoclastogenesis process during bacterial-mediated osteolytic infections. Therefore, to target LPS-induced osteoclastogenesis in inflammatory bone loss in diseases such as periodontitis, rheumatoid arthritis, etc., future investigations will use proper animal model systems to determine C-PC's effect on bone loss in vivo.
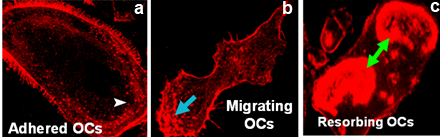 Figure 1: Confocal images of osteoclasts demonstrating podosomes at the cell periphery and leading edge (a &b); resorbing OC demonstrates sealing rings. |
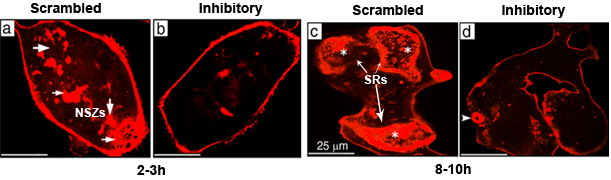 Figure 2: Confocal images of osteoclasts (OCs) plated on a mineralized matrix and stained with rhodamine-phalloidin. Actin distribution is shown in OCs incubated with scrambled (a & c) and inhibitory (b & d) LPL peptides in the presence of TNF-α. At the indicated time, OCs incubated with LPL inhibitory peptide failed to exhibit either NSZs (b) or sealing rings (d). Scale bar-25 µm. |
- Project 3- Differentiation of stem cells to osteoblasts and bone formation- Analysis of the effect of Methylsulfonylmethane (MSM) in bone remodeling
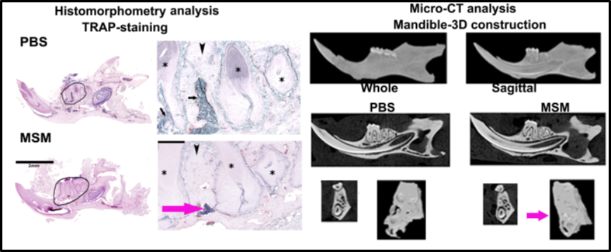
Methylsulfonylmethane (MSM) is a sulfur compound with an anti-inflammatory effect. Stem cells and their role in bone remodeling in the presence of MSM is another ongoing project developed by Dr. Chellaiah and a graduate student (Dr. Hanan AlJohani, who graduated in 2020). Studies with stem cells from human exfoliated deciduous teeth (SHED) provided essential insight into the effect of MSM on bone formation in vitro. In vivo experiments in aging mice, MSM increased bone formation in bone loss areas observed in the inter-radicular region (Fig.4; pink arrows). Immunohistochemical analyses of the mandibles for the osteoblast-specific marker osteocalcin (OCN) and the mesenchymal stem cell-specific marker CD105 showed a significant increase in OCN and a decrease in CD105-positive cells in response to MSM injection in aging mice. MSM seems to reduce osteoclast function and not differentiation, which will be one of the highlights of the MSM effect.
Current studies focus on the effect of MSM on stem differentiation in vitro and in vivo. The usage of MSM may ultimately impact the treatment of bone-loss diseases associated with inflammation, including periodontitis and rheumatoid arthritis, which share several pathogenic features with osteoporosis.
- Projects (4 and 5) in Prostate cancer (PCa)
4. CD44-ICD and transcriptional regulation of RUNX2: Prostate cancer (PCa) is the second leading cause of cancer-related death in men in the United States, partially due to metastatic spread to secondary sites in the bone, brain, lymph nodes, and visceral organs. Prostate cancer metastasizes to bone and produces osteoblastic or osteolytic lesions. Intracardiac injection of PCa cells (PC3) into nude mice demonstrated the spread of PC3 cells to bone. It produced osteolytic lesions with the differentiation and activation of osteoclasts. CD44 is a cell surface receptor with extracellular (ECD), transmembrane (TMD), and intracellular (ICD) domains. Dr. Linda Senbanjo's thesis depicted cloning a series of C-terminal deletion constructs and chromatin immunoprecipitation assays that the CD44-intracellular domain (ICD) has RUNX2 binding sites between amino acids 671 and 706. Results identified that the sequence-specific interaction of CD44-ICD with RUNX2 has a functional relationship to the transcription of metastasis-related genes, e.g., MMP9 and RANKL. Her results allude to CD44-ICD as a novel therapeutic target in cancer cells that express CD44. Future studies will focus on the function of CD44-ICD on metastasis-related events.
5. L-plastin and Invasion: In the other project, the Chellaiah laboratory (Characterized by Dr. Majumdar, a post-doctoral fellow) determined the role of L-plastin (LPL) in PCa cell (PC3 cells) invasion via invadopodia formation. Knockdown of LPL in PC3 cells diminished invasion by reducing invadopodia formation. L-plastin is emerging as a novel therapeutic target due to its cell-specific activation in cancer cells and osteoclasts, not in other normal cells.
Peer-Reviewed Publications Pertinent to the Projects
Complete List of Published Work in My Bibliography: My Bibliography - NCBI (nih.gov)
Dr. Chellaiah's research outcomes, a testament to her dedication and expertise, have been published in prestigious, peer-reviewed scientific journals, which indicates her work's high quality and impact. These publications highlight her work's significance and contribute to advancing understanding of the field.
- Aljohani H, Senbanjo LT, Al Qranei M, Stains J, and Chellaiah MA. Methylsulfonylmethane increases the alveolar bone density of mandibles in aging female mice. Front. Physiol. 12:708905. doi 10.3389/fphys. 2021.Oct.4; 708905
- Chellaiah MA. L-Plastin Phosphorylation: Possible Regulation by a TNFR1 Signaling Cascade in Osteoclasts. Cells 2021 Sept 9; 10(9), 2432; https://doi.org/10.3390/cells10092432
- Majumdar S, Senbanjo LT, AlJohani H, Chellaiah MA. L-plastin regulates invasion and possibly not the migration of prostate cancer (PC3) cells. Journal of Cancer Research and Therapeutic Oncology 9: 1-17.
- Aljohani H, Stains JP., Majumdar S, Srinivasan D, Senbanjo L, Chellaiah MA.Peptidomimetic inhibitor of L-plastin reduces osteoclastic bone resorption in aging female mice. Bone Res 2021 Apr 9; 9: 22 (2021). https://doi.org/10.1038/s41413-020-00135-9.
- AlQranei MS, Senbanjo LT, Aljohani H, Hamza T, Chellaiah MA.Lipopolysaccharide- TLR-4 Axis regulates osteoclastogenesis independent of RANKL/RANK signaling. BMC Immunol. 2021 Mar 25;22(1):23. doi: 10.1186/s12865-021-00409-9—pubMed PMID: 33765924; PubMed Central PMCID: PMC7995782.
- Senbanjo LT, AlJohani H, AlQranei M, Majumdar S, Ma T, Chellaiah MA. Identification of sequence-specific interactions of the CD44-intracellular domain with RUNX2 in the transcription of matrix metalloprotease-9 in human prostate cancer cells. Cancer Drug Resist. 2020 3rd Quarter;3(3):586-602. doi: 10.20517/cdr.2020.21. Epub 2020 Aug 21. PubMed PMID: 33062960; PubMed Central PMCID: PMC7556329.
- Chellaiah MA. Osteoclast Cytoskeleton, Podosome, Motility, Attachment, and Signaling by Receptors.Encyclopedia of Bone Biology: Zaidi, M (ed.) Oxford: Academic Press. 2020 July; 1:236-250.
- AlQranei MS, Chellaiah MA. Osteoclastogenesis in periodontal diseases: Possible mediators and mechanisms. J Oral Biosci. 2020 Jun;62(2):123-130. doi: 10.1016/j.job.2020.02.002. Epub 2020 Feb 17. Review. PubMed PMID: 32081710; PubMed Central PMCID: PMC7329621.
- AlQranei MS, Aljohani H, Majumdar S, Senbanjo LT, Chellaiah MA. C-phycocyanin attenuates RANKL-induced osteoclastogenesis and bone resorption in vitro by inhibiting ROS levels, NFATc1, and NF-κB activation. Sci Rep. 2020 Feb 13;10(1):2513. Doi: 10.1038/s41598-020-59363-y—PubMed PMID: 32054921; PubMed Central PMCID: PMC7018981.
- Chellaiah MA, Moorer MC, Majumdar S, Aljohani H, Morley SC, Yingling V, Stains JP. L-plastin deficiency produces increased trabecular bone due to attenuation of sealing ring formation and osteoclast dysfunction. Bone Res. 2020; 8:3. Doi: 10.1038/s41413-019-0079-2. eCollection 2020. PubMed PMID: 31993243; PubMed Central PMCID: PMC6976634.
- Aljohani H, Senbanjo LT, Chellaiah MA. Methylsulfonylmethane increases osteogenesis and regulates the mineralization of the matrix by transglutaminase 2 in SHED cells. PLoS One. 2019;14(12): e0225598. doi: 10.1371/journal.pone.0225598. eCollection 2019. PubMed PMID: 31805069; PubMed Central PMCID: PMC6894810.
- Senbanjo LT, Chellaiah MA. CD44 cleavage product CD44-intracellular domain regulates gene transcription and tumorigenesis. Cancer Studies and Therapeutics. 2019 September; 4(4):1-2.
- Senbanjo LT, AlJohani H, Majumdar S, Chellaiah MA. Characterization of CD44 intracellular domain interaction with RUNX2 in PC3 human prostate cancer cells. Cell Commun Signal. 2019 Jul 22;17(1):80. Doi: 10.1186/s12964-019-0395-6—PubMed PMID: 31331331; PubMed Central PMCID: PMC6647163.
- Majumdar S, Wadajkar AS, Aljohani H, Reynolds MA, Kim AJ, Chellaiah MA. Engineering of L-plastin Peptide-Loaded Biodegradable Nanoparticles for Sustained Delivery and Suppression of Osteoclast Function In Vitro. Int J Cell Biol. 2019; 2019:6943986. Doi: 10.1155/2019/6943986. eCollection 2019. PubMed PMID: 31191656; PubMed Central PMCID: PMC6525930.
- Homayounfar N, Khan MM, Ji Y, Khoury ZH, Oates TW, Goodlett DR, Chellaiah M, Masri R. The effect of embryonic origin on the osteoinductive potential of bone allografts. J Prosthet Dent. 2019 Apr;121(4):651-658. doi: 10.1016/j.prosdent.2018.09.003. Epub 2018 Dec 28. PubMed PMID: 30598313.
- Chellaiah MA, Ma T, Majumdar S. L-plastin phosphorylation regulates the early phase of sealing ring formation by an actin-bundling process in mouse osteoclasts. Exp Cell Res. 2018 Nov 1;372(1):73-82. doi 10.1016/j.yexcr.2018.09.014. Epub 2018 Sep 21. PubMed PMID: 30244178; PubMed Central PMCID: PMC6181588.
- Chellaiah MA, Majumdar S, Aljohani H. Peptidomimetic inhibitors of L-plastin reduce the resorptive activity of osteoclasts but not the bone-forming activity of osteoblasts in vitro. PLoS One. 2018; 13 (9): e0204209. Doi: 10.1371/journal.pone.0204209. eCollection 2018. PubMed PMID: 30248139; PubMed Central PMCID: PMC6152981.
- Srinivasan D, Senbanjo L, Majumdar S, Franklin RB, Chellaiah MA. Androgen receptor expression reduces stemness characteristics of prostate cancer cells (PC3) by repressing CD44 and SOX2. J Cell Biochem. 2018 Sep 11; 120(2):2413-2428. doi: 10.1002/jcb.27573. [Epub ahead of print] PubMed PMID: 30206982; PubMed Central PMCID: PMC6411465.
- Costello LC, Chellaiah MA, Zou J, Reynolds MA, Franklin RB. In vitro, BMP2 stimulation of osteoblast citrate production coincides with mineralized nodule formation. J Regen Med Tissue Eng. 2015; 4. pii: 2. Epub 2015 Oct 17 [PMCID: PMC4666534]
- Franklin RB, Chellaiah MA, Zou J, Reynolds MA, Costello LC. Evidence that osteoblasts are specialized citrate-producing cells that provide citrate for incorporation into the structure of bone. The Open Bone J. 2015 (Dec) 6:1-7 [PMCID: PMC4346336]
Alums of the Chellaiah Lab
- Post-doctoral fellows (2000 to 2020)
Rajat Biswas, Ph.D.
Venkatasababa Samanna Ph.D.
Tao Ma, Ph.D.
Aditi Gupta, Ph.D.
Vinodh Rajagopalan Ph.D.
Sunipa Majumdar Ph.D.
Linda Senbanjo, Ph.D.
- Research Fellow (2016-2018)
Deepa Srinivasan MS
- Graduate students (2004-2020)
(graduated years in parenthesis)
Bhavik Desai (2008)
Brian Robertson (2010)
Linda Senbanjo (2019)
Hanan Aljohani (2020)
Mohammed AlQranei (2020)
- Research Assistants
David Yuen (2000-2002)
Kavitha Sadashivaiah (2009)
- Exchange Visitors
Ganglao Gao, a physician from China (2013)
Lei Lei (China; 2012)
Han Ji Young (Korea; 2012)
Hongzhi Zhou (Physician and Professor from China, Came on Sabbatical in 2011)
Wei Cao (Ph.D. candidate from Dr. Wantao Chen's laboratory, China; 2010)
Employment History
| April 2024 – Present | Professor, Department of Biomaterials and Regenerative Dental Medicine Greenebaum Comprehensive Cancer Center |
| August 2014-Present | Professor, Department of ODS; SOD, University of Maryland, Baltimore |
| 2007 Aug-July 2014 | Associate Professor, Department of ODS; SOD, University of Maryland, Baltimore |
| July 2000-2007 | Assistant Professor, Department of BMS; SOD, University of Maryland, Baltimore |
| 1999-June 2000 | Research Assistant Professor, Department of Medicine; Washington University School of Medicine, St. Louis, Missouri |
Honors and Awards
| 2009-2010 | American Society for Bone and Mineral Research Award ($50,000) |
| 1999-2000 |
Barnes-Jewish Research Foundation, St. Louis, MO $50,000 |
| 1996-1997 | Barnes-Jewish Research Foundation, St. Louis, MO $50,000 |
| 1991-1992 | NIADDK training grant award |
| 1989-1990 | NRS training grant award |
| 1980-1985 | Council of Scientific and Industrial Research Award for Ph.D. program |
| 1979 | University IV Rank Holder in MSc (India) |
| 1977-1979 | Awarded National Merit Scholarship for Master program (MSc) in India |
| 1977 | University II Rank Holder in bachelor's degree (BSc) for which National Merit Scholarship was awarded (India) |
Completed Grants
- Proposal Title - L-Plastin: A Novel Target for Intervention in the Treatment of Osteoporosis. Principal Investigator. NIH/NIAMS. NIH R01 AR066044-01 (09/15/2014-08/30/2019). No cost Extension until 2021
- Proposal title- Regulation of signaling in Osteoclast Bone Resorption Principal Investigator. NIH/NIAMS.R56AR046292-09A1(07/01/2009 to 06/30/2013)
- Proposal title- Regulation of signaling in Osteoclast Bone Resorption Principal Investigator. NIH/NIAMS. NIH 2R01 AR046292-05A2 (09/15/2004-08/30/2009)
- Bridge Funding Award (2008). Principal Investigator. Agency- American Society for Bone and Mineral Research
- Proposal title- Gelsolin-based signaling in Osteoclast function. Principal Investigator. NIH/NIAMS. NIH 2R01 AR046292-05A2 (09/15/1999-08/30/2003)
- Proposal title: The Role of Osteopontin in Cancer Progression and Metastasis F30 Student Grant
Role-Mentor. Brian Robertson (Principal investigator/ graduate student in DDS/Ph.D. program). NIH-NIDCR. 1 F30 DE018308-01 (06-01-07 to 06-31-09).