contact
Shereece Singleton
Office Manager
F 410-706-0865
Lab Summary
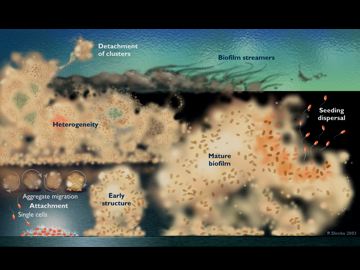 A biofilm may be defined as a microbially-derived sessile community characterized by cells that are attached to a substratum, interface, or to each other, are embedded in a matrix of extracellular polymeric substances, and exhibit an altered phenotype with respect to growth, gene expression, and protein production. This layer prevents infection resolution by antimicrobial agents and host phagocytic cells. Once a biofilm is formed on host tissue or an indwelling medical, a chronic infection ensues and often the only treatment option is debridement or implant removal.
A biofilm may be defined as a microbially-derived sessile community characterized by cells that are attached to a substratum, interface, or to each other, are embedded in a matrix of extracellular polymeric substances, and exhibit an altered phenotype with respect to growth, gene expression, and protein production. This layer prevents infection resolution by antimicrobial agents and host phagocytic cells. Once a biofilm is formed on host tissue or an indwelling medical, a chronic infection ensues and often the only treatment option is debridement or implant removal.
My laboratory is presently using 2D gel electrophoresis, microarray analysis, reporter systems, and knockouts to identify biofilm specific genes and their products in Staphylococcus aureus, Proteus mirabilis, Acinetobacter baumannii, and Klebsiella pneumoniae. In a long standing collaboration with Dr. Jeff Leid's lab in Northern Arizona University as well as with Dr. Jason Calhoun at Ohio State University Medical School, we are using these specific products to design vaccines that protect against biofilm infections using several animal models. In addition, we are developing tagged monoclonal antibodies against biofilm specific proteins for a rapid, sensitive, and specific diagnosis of endocarditis, prosthetic implant infection, deep abscess, and osteomyelitis.
|
2011 Nature Medicine Article on Shirtliff Lab |
Another aspect of our research involves understanding the interaction of different microbial species that coexist within complex polymicrobial biofilms in collaboration with Dr. Mary Ann Jabra-Rizk in the Department of Oncology and Diagnostic Sciences. While the fungal species Candida albicans and the bacterial species S. aureus are responsible for the majority of hospital-acquired infections and are often co-isolated in a biofilm mode of growth, these microbes also demonstrate infectious synergism. We are presently exploring the microscopic and molecular interactions between these two microbial species using confocal scanning laser microscopy, microarray, and differential 2D-based proteomic approaches. In addition to this dual species biofilm infection, we are also interested in the many microbial species that interact in biofilms in infectious diseases including cystic fibrosis, chronic wounds, polymicrobic musculoskeletal infections, and catheter (urinary and intravenous) catheter infections.
---
Following Professor Mark Shirtliff’s death on July 12, 2018, his long-term research collaborator and lab manager, Dr. Janette Harro, is continuing most of the projects described on this web page. Please address all queries to Dr. Harro at jharro@umaryland.edu. This web page will be updated in due course.
